Iodine Derivatives
- Methyl Iodide
- Ethyl Iodide
- n-Butyl Iodide
- N-propyl Iodide
- 2-Ethylhexyl Iodide
- Diiodomethane
- Chloroiodomethane
- 5-Iodo-2-methylbenzoic Acid
- 2-chloro-5-iodobenzoic acid
- Methyl-2-Iodobenzoate
- 2-Iodobenzoic Acid
- 3,5-Diiodosalicylic Acid
- N-Iodosuccinimide
- Tetrabutylammonium Iodide
- Iodine Monochloride
- 5-Iodoanthranilic acid
- Iodoform
- Potassium iodide
- Sodium Iodide
- Ammonium Iodide
- Cuprous Iodide
- Zinc Iodide
- Sodium Metaperiodate
- Potassium Iodate
- Calcium Iodate
- Iodic acid
- Periodic acid
- Hydriodic Acid
- Iodobenzene Diacetate
- Dess Martin Periodinane
- 1,4-Diiodobenzene
- 4-Bromoiodobenzene
- 4-Chloroiodobenzene
- Iodobenzene
- 4-Iodoaniline
- 4-Iodotoluene
- 4-Iodoanisole
- 4-Iodophenol
- 4,4'-Diiodobiphenyl
- Trimethylsulfoxonium Iodide
Ethyl Iodide (Iodo Ethane)
Ethyl iodide is commonly known as Iodo ethane, having Molecular formula C2H5I, Mol. Wt.155.97 g/mol.It is a Primaryhalogeno- alkane compound in which one Hydrogen atom in an alkane (like Ethane) is replaced by a halogen atom (ie. Iodine atom). It represents the category of Organo iodide compounds, with the presence of one Carbon–Iodine bond. CAS No. of Ethyl Iodide is 75-03-6.
It can be prepared by any one of the following methods:
- By the action of Iodine on Alcohol with Red Phosphorous
- From Diethyl ether and Hydriodic acid
Ethyl iodide is a dense, colorless transperrent, flammable liquid . It is sensitive to light and on exposure to light turns slight reddish due to liberation of Iodine. It has a boiling point (B.P. 72.3-72.4 °c), Sp. Gr. at 20°C; 1.9 –1.93 gm/cc. It is slightly soluble in water, [solubility in water 0.4 g/100ml at 20 °c], it is completely miscible with Ethanol.
Ethyl iodide is stored in dark amber colored glass bottles to prevent its degradation by light. It is stabilized by Copper or Silver wire. It needs to be handled carefully as it is harmful by inhalation, or if swallowed or through skin contact. It is used mainly as an ethylating agent in Organic synthesis.
| Product | Ethyl Iodide (Iodo Ethane) |
| Structural formula | 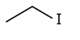 |
| CAS No. | [75-03-6] |
| Molecular formula | C2H5I |
| Molecular weight | 155.97 |
| Description | Clear colorless liquid |
| Density | Between 1.90 and 1.93 g/ml. |
| Water | Max. 0.50 % |
| Assay | Min. 99.0 %. |